Fundamentals of Corrosion
Introduction
To understand the Corrosion, first we need to understand some fundamentals of corrosion. Corrosion is usually described by its results. The terms rust, scaling, discoloration, oxidation, pitting, etc., are familiar terms. These descriptive terms focus on the readily observable characteristics of corrosion products, which are results of the corrosion process.
The corrosion process acts upon engineered materials, usually metals. Engineered materials are produced by man to serve as components of society’s infrastructure. For the purpose of this discussion, steel represents
the most common material used in industrial and marine construction. Steel is composed principally of approximately 95% iron (Fe). Most of the economically significant corrosion in industry results from the deterioration of iron.
Corrosion is not only dangerous; it is also an extremely costly problem that impacts nearly every industry. You should be aware that what are the impact of corrosion.
Read this article for more details…The Impact of Corrosion
Definition
“The corrosion process is the deterioration of a substance, usually a metal, or its properties, because of a reaction with its environment.”
Corrosion Process
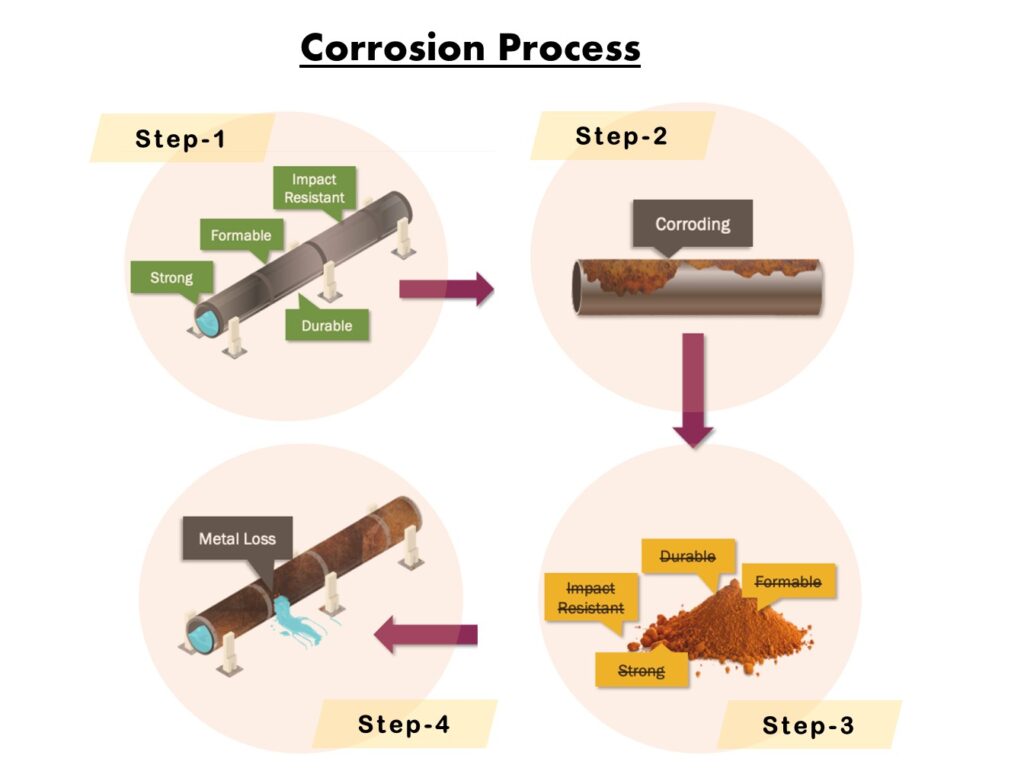
There is a basic law of nature that states that the more energy you add to a substance to change it into something else (e.g., iron ore to steel), the faster it wants to return to its original low energy state. The corrosion process generally divided in to 4 steps.
- Step-1 Metal to be present
- Step-2 Metal to be react
- Step-3 Transformation of the metal.
- Step-4 Deterioration of the metal.
The Corrosion Process: Step 1
The first step in the metallic corrosion process requires metal to be present. A diverse range of metals and alloys are susceptible to corrosion, and steel is no exception. It should be noted that steel actually describes an entire family of metal alloys, with hundreds of application-specific grades.
Carbon steel, as it is commonly used within the construction industry. Steel is an alloy made out of iron ore and carbon and, its properties are chiefly dependent on the percentage of carbon present. Carbon steel contains a carbon content of up to 2.1% by weight. Other elements may be added but in smaller quantities, often too small to affect its properties.
The Corrosion Process: Step 2
In the second step in the corrosion process, the metal reacts physically and chemically with its environment. When iron or steel corrodes, the result is iron oxide, or what we call rust. The term rust refers to the oxidation of iron. Oxidation, or rusting occurs as a result of the chemical reaction between steel and oxygen that requires the simultaneous presence of water and oxygen. Oxygen is always present, even when metal is buried under the ground or submerged in water. Likewise, water is present in most environments. Just a few potential sources of water include rain, snow, condensation, humidity, and steam.
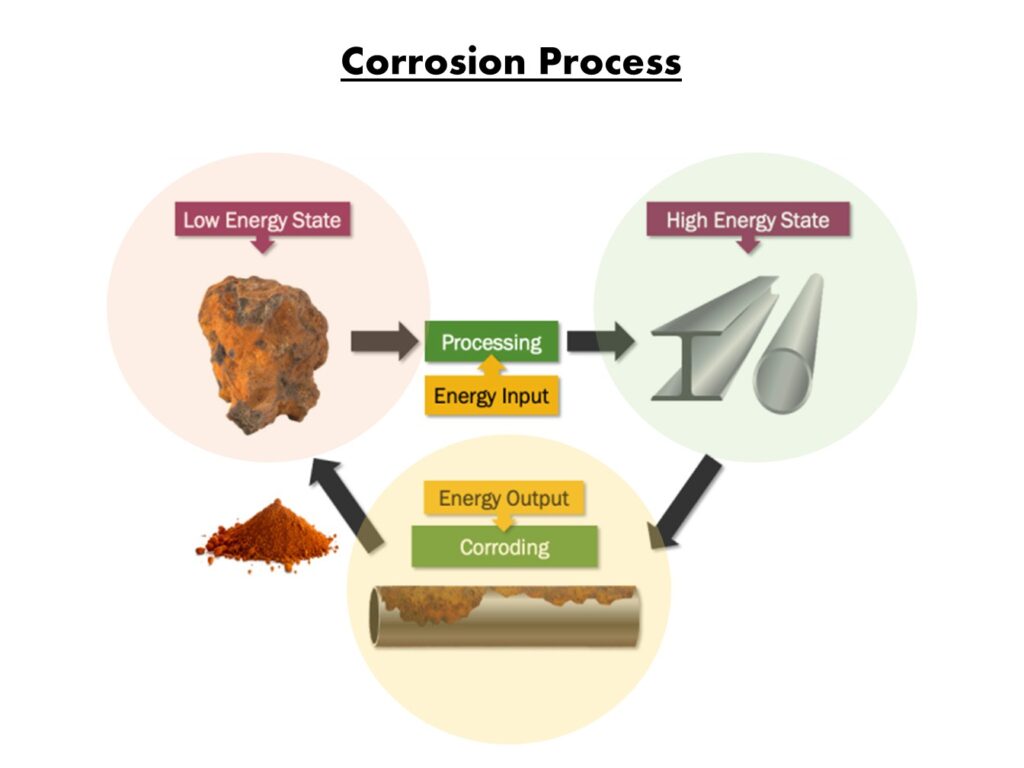
When iron is exposed to water and oxygen, it reacts in an attempt to return to its natural state. This happens because carbon steel is not a naturally occurring material and must be manufactured in a foundry. Steel is created by processing iron ore and then forging the ore with carbon and other elements, as discussed in Step
1. During the processing of the iron ore, a tremendous amount of energy is added in the form of heat, increasing the steel’s energy level well above the original iron ore’s baseline energy state.
A diverse range of metals and alloys are susceptible to corrosion, and steel is no exception. Carbon steel is made up of approximately 97% iron ore; it takes a tremendous amount of energy to change the ore to steel; consequently, nature will rapidly begin to change steel back into iron oxide (rust) quickly through the corrosion process.
The Corrosion Process: Step 3
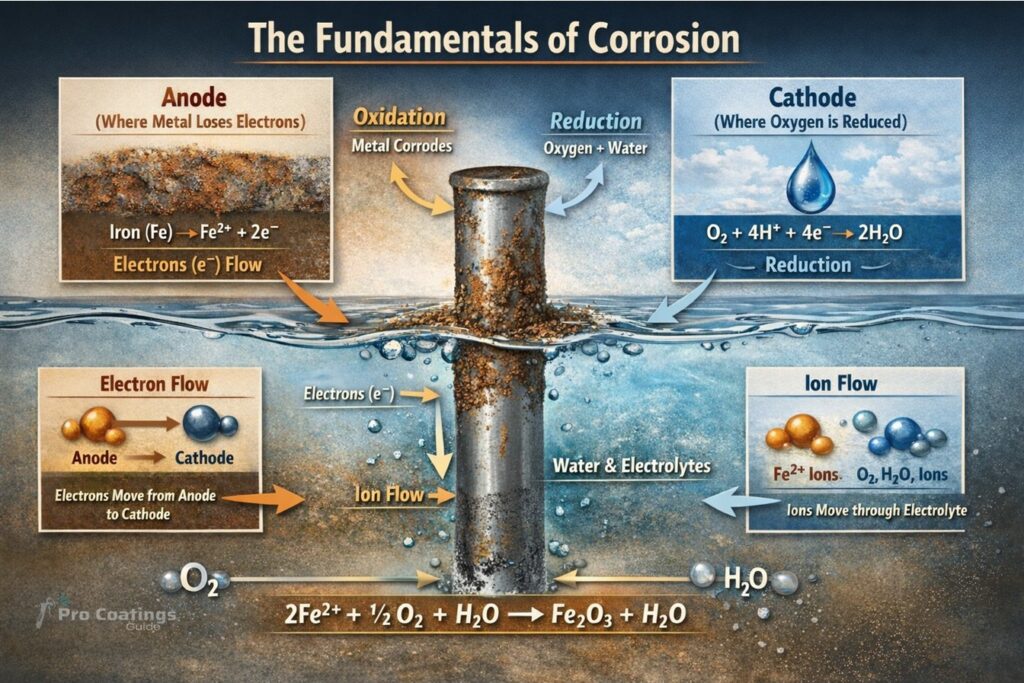
The third step in the corrosion process involves the transformation of the metal into other chemical compounds. The transformation occurs through the formation of an electrochemical cell and the subsequent reactions. These reactions result in the metal breaking down and forming iron oxide.
In order for the metal to transform and corrode, two key reactions occur simultaneously within the electrochemical cell. At the anode, the metal is oxidized, causing it to lose mass and dissolve into the electrolyte. This process generates excess electrons at the anode, which then travel to the cathode. At the cathode, reduction takes place, and the electrons are consumed. During the corrosion process, the reactions between the anode, cathode, and electrolyte combine with oxygen to complete the corrosion process and produce iron oxide, otherwise known as rust.
It is important to highlight that in the real world, electrochemical cells do not exist in isolation. Instead, there are millions of anodic and cathodic sites across the surface of the steel that are simultaneously reacting. If you look at the image on the slide you can see this process. The anodic sites are where the metal has been eaten away or oxidized. The cathodic sites are where the reduction has taken place and the corrosion product, iron oxide, has formed.
Electrochemical Cell (Corrosion Cell )
An electrochemical cell, sometimes referred to as a corrosion cell or corrosion circuit, is a condition on a metal surface where electric current flows between two metal surfaces. Electrochemical cells can be broken down into four functional components.
- Anode
- Cathode
- Metallic Pathway
- Electrolyte
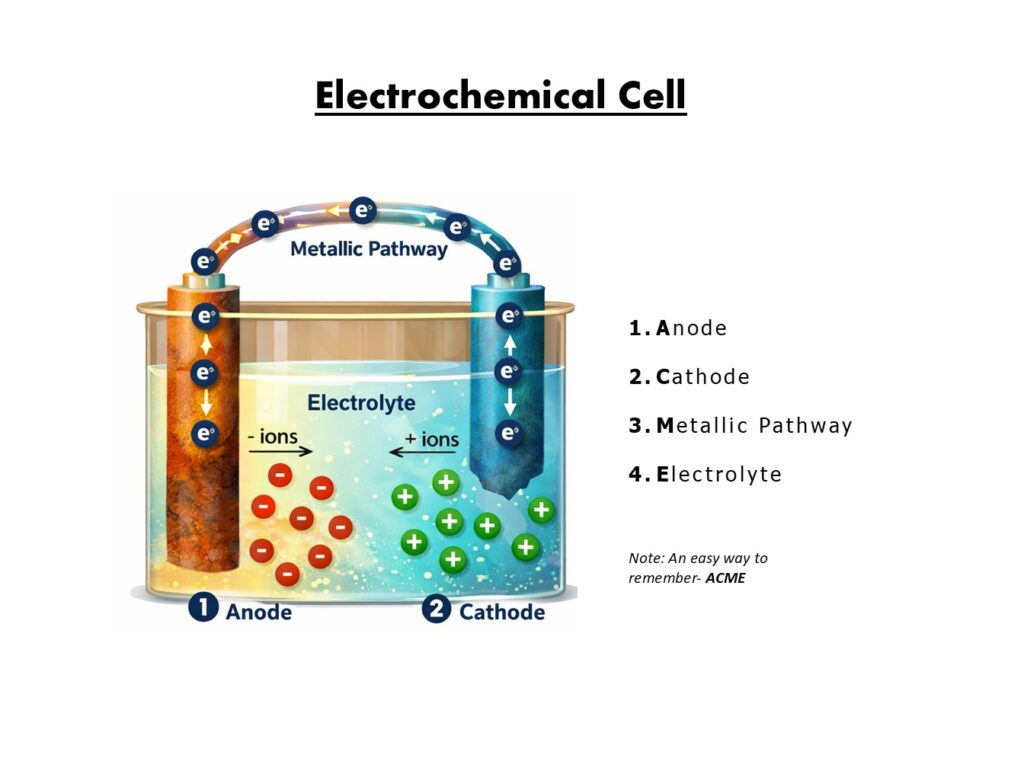
1. Anode
The anode is the area of the metal surface that corrodes. The metal oxidizes (gains oxygen), causing it to lose mass and dissolve into the electrolyte. The metal that dissolves does so in the form of positively charged cations that then migrate to the cathode through the electrolyte.
Oxidation: A chemical reaction that occurs at the anode where the metal loses electrons.
2. Cathode
The cathode is the area of the metal surface that reduces (loses oxygen), does not corrode and is protected. The metal forms negatively charged anions that migrate to the anode through the electrolyte. Electrons flow from the anode toward the cathode in the metallic pathway.
Reduction: A chemical reaction that occurs at the cathode that involves the gain of electrons to the
metal. Reduction and oxidation always occur together through an exchange of electrons at specific
sites on the metal surface.
3. Metallic Pathway
The metallic pathway connects the anode and cathode and allows the passage of electrons generated at the anode to the cathode. The metallic pathway is often the underlying metal substrate.
4. Electrolyte
The electrolyte is a conductive medium in which ions are carried from the anode to the cathode. The electrolyte is any solution capable of conducting electricity, such as water.
All four of the components of the electrochemical cell (anode, cathode, return path, and electrolyte) must be present for corrosion to occur. Removing one or more of them prevents corrosion from occurring. As you can imagine, it is not always possible or practical to remove these components, but the attempt to remove them is called corrosion control.
The Corrosion Process: Step 4
The fourth and final step in the corrosion process involves the degradation of the metal. Steel and other metals are highly susceptible to corrosion. This is the result of the iron reverting back to its natural, more stable iron oxide. As the iron oxide produced in the corrosion process consumes the metal, the components of the steel that provide the beneficial properties such as strength, durability, hardness, and impact resistance that were created during the refining process are lost. In addition, the volume and thickness of steel is also reduced. These factors lead the steel to lose structural integrity. Depending on the function of the metal, its impairment can be disastrous.
For more understanding of the corrosion and it’s control, we need to understand the types of corrosion and factors that affects the rate of corrosion.
Reference and Standards:
- AMPP (The Association for Materials Protection and Performance)
- SSPC (Society for Protective Coatings).
