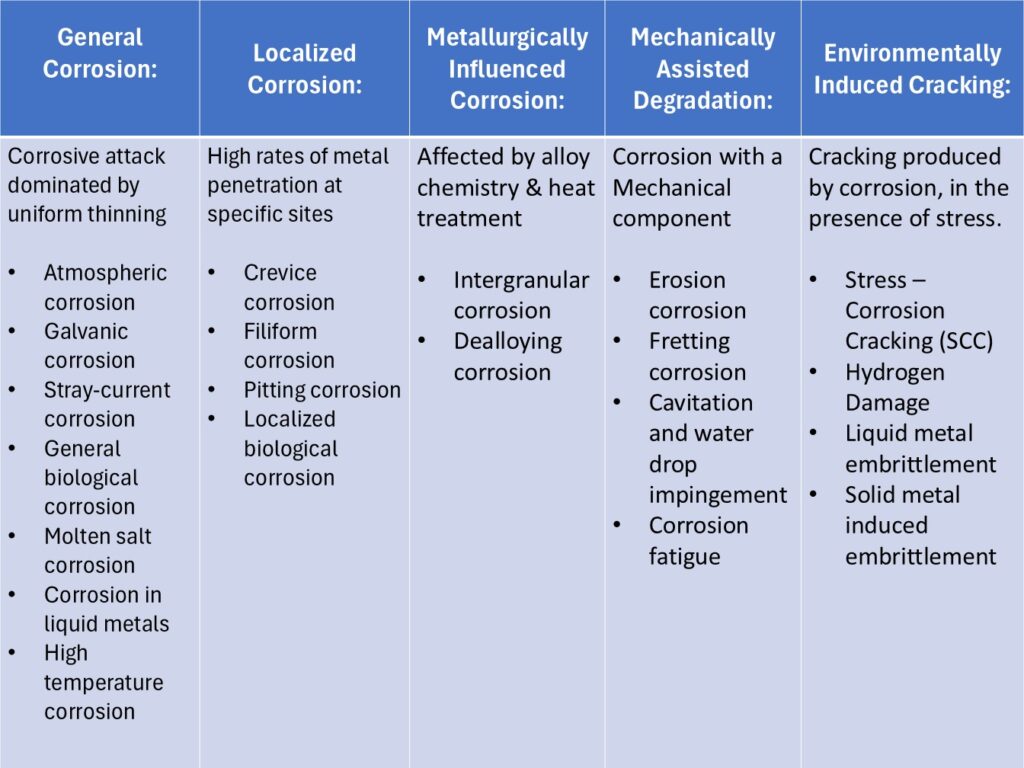
Types of Corrosion

Types of Corrosion
Even though the fundamentals of corrosion involves creation or existence of corrosion cells, there are several types of corrosion that can occur. It should however be borne in mind that for corrosion to occur, there is no need for discrete (physically independent) anodes and cathodes. Innumerable micro level anodic and cathodic areas can be generated at the same (single) surface on which anodic (corrosion) and cathodic (reduction) reactions occur.
Each types of corrosion has a specific arrangement of anodes and cathodes and specific patterns and locations depending on the type can exist.
The most important types are
- Uniform corrosion.
- Galvanic corrosion, concentration cells, water line attack
- Pitting.
- Dealloying (selective leaching)
- Atmospheric corrosion.
- Erosion corrosion
- Fretting
- Crevice corrosion; cavitation
- Stress corrosion,
- Intergranular and transgranular corrosion,
- Hydrogen cracking and embrittlement
- Corrosion fatigue.
Classifications of corrosion types
General/Uniform Corrosion:
General types of corrosion results in a relatively uniform loss of material over the entire surface and in a general thinning of the affected surface. General corrosion is relatively easy to visually inspect and very rarely contributes to catastrophic failures. Nondestructive testing is generally required to determine losses.
Localized Corrosion:
- In addition to General types of corrosion, there is also some localized corrosion occurred.
- Localized corrosion occurs at discrete sites on the metal surface and is typically confined to a particular area. As a result, the areas immediately adjacent to the localized corrosion normally corrode much slower, if at all.
- Localized corrosion can be difficult to inspect as access to these discrete sites can be challenging. This form of corrosion is less common in atmospheric exposure and more common in immersion, splash, or spray
Galvanic Corrosion
- Galvanic/Dissimilar Corrosion occurs when two metals/alloys with different potentials come into contact within a corrosive electrolyte.
- Rate of attack of the more active metal is usually accelerated, while corrosion of the more noble metal is decreased or stopped.

Crevice Corrosion
- Crevice Corrosion occurs on a metal surface that is shielded from full exposure to the environment, and because of the proximity of another material that forms a narrow gap between them.
- Common in situations where there is metal-to-metal contact, such as in support straps or at pipe flanges.

Pitting Corrosion
- Pitting Corrosion occurs at distinct spots where deep pits are produced. Pitting can be isolated, or a group of pits may grow together to form a single larger pit.
- Typically caused by overcoating contaminated surfaces that contain nonvisible contaminants such as
chlorides, sulfates, and nitrates. - Especially prevalent in metals that form a protective oxide layer and in environments high in chloride
concentration

Beyond the most common types of degradation, corrosion can manifest in more specialized and often more destructive ways depending on the environment and the mechanical stresses involved. Let’s disscuss on some specialized types of corrosion.
Stress Corrosion Cracking (SSC)
- Stress corrosion cracking (SCC) refers to failure under simultaneous presence of a corrosive medium and tensile stress.
- Unlike general corrosion, where a metal simply rusts away uniformly, SCC is localized. It often starts at a microscopic pit or a scratch on the surface.
- Once the crack starts, the stress at the tip of that crack becomes incredibly high. This prevents the metal from healing its protective layer, allowing the chemicals to continue eating away at the material deep inside the crack.
- The crack grows often following grain boundaries until the remaining solid metal can no longer support the load, leading to a sudden, often catastrophic, break

- The above image shows the failure of a water pipeline due to SCC. High temperatures accelerate the SCC process by enhancing the rate of corrosion and weakening the mechanical properties of the metal, making it more prone to cracking under stress.
- As the temperature increases, the metal’s microstructure can also change, making it more susceptible to cracking in the presence of corrosive agents like chlorides or sulfides.
Intergranular Corrosion
- Intergranular corrosion is an attack on or adjacent to the grain boundaries of a metal or alloy.
- A highly magnified cross section of most commercial alloys will show its granular structure. This structure consists of quantities of individual grains, and each of these tiny grains has a clearly defined boundary that chemically differs from the metal within the grain center.
- Heat treatment of stainless steels and aluminum alloys accentuates this problem.

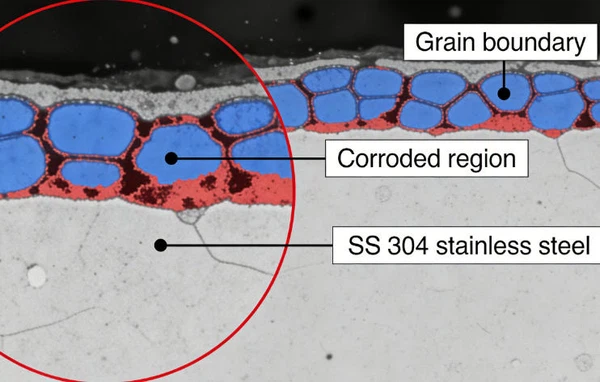
- The picture above shows a stainless steel which corroded in the heat affected zone a short distance from the weld. This is typical of intergranular corrosion in austenitic stainless steels.
Erosion Corrosion
- Erosion is one of the most aggressive mechanisms of deterioration in hydraulic and industrial equipment, due to the relative movement of a corrosive fluid with respect to the surface of the material.
- The protective surface films are continuously removed, exposing fresh metal to corrosive attack. It commonly affects piping, pump impellers, elbows, and heat exchanger tubes.

Fretting corrosion
- Fretting corrosion occurs at contact surfaces subjected to small, repeated relative movements, such as vibration or oscillation. These micro-motions damage protective oxide layers, leading to oxidation and material degradation.
- Fretting is often observed in bolted joints, bearings, couplings, and mechanical assemblies.

Cavitation
- Cavitation is a particular type of erosion that occurs when the operating pressure of a fluid falls below its vapor pressure, causing the formation of vapor bubbles that collapse violently on the metal surface. This creates shock waves and damage the metal surface.
- This often affects pumps, propellers, and hydraulic equipment.

Corrosion Fatigue
- While stress corrosion cracking involves a static load, corrosion fatigue is the result of the combined action of an alternating (cyclic) stress and a corrosive environment.
- The corrosive environment creates tiny pits or notches on the metal surface. As the component bends, vibrates, or rotates, the cyclic stress pulls these pits apart.
- The corrosive agent (like saltwater or acid) then rushes into the fresh crack, preventing the metal from healing its protective oxide layer.
- The crack grows deeper with every cycle until the remaining solid metal can no longer support the load, leading to a sudden, catastrophic snap.
- This type of corrosion is common in rotating equipment, offshore structures, aircraft components, and bridges.

Microbiologically Influenced Corrosion (MIC)
- Microbiological Influenced Corrosion (MIC) is a fascinating if slightly unsettling process where living organisms actually eat or chemically attack engineering materials.
- It isn’t a unique form of corrosion itself, but rather a biological accelerator that uses microbes to trigger or speed up the electrochemical reactions we usually see in pitting or crevice corrosion.
- MIC is typically caused by colonies of bacteria, algae, or fungi that attach to a surface and form a biofilm. This slimy layer creates a localized “micro-environment” that is chemically different from the rest of the fluid. This leads to rapid, deep pitting that can breach a thick steel pipe in a fraction of the time predicted by standard corrosion models.
- This type of corrosion found generally in Oil and gas pipelines, water treatment facilities and fire sprinkler systems.

Hydrogen Embrittlement
- It is a process where metals, particularly high-strength steels, become brittle and fracture due to the introduction and diffusion of atomic hydrogen into the crystal lattice.
- Atomic hydrogen is small enough to slip between the larger metal atoms. It can be introduced during manufacturing (like electroplating or welding) or as a byproduct of other corrosion reactions
- Once inside, the hydrogen atoms migrate through the metal. They tend to follow stress gradients, gathering at the tips of tiny internal cracks or at grain boundaries.
- There are several theories on what happens next, but the result is the same: the hydrogen interferes with the metal’s ability to slip or deform plastically. Instead of bending under a load, the metal becomes glass-like and snaps.

Understanding the different types of corrosion helps engineers and inspectors identify damage early, prevent structural failures, and reduce maintenance and repair costs. It also helps to choose and understand effective corrosion control methods.
In the next articles, we will discuss on different corrosion control methods include proper material selection, application of protective coatings, cathodic protection systems, corrosion inhibitors, and control of environmental factors such as moisture and contaminants.


