Table of Contents
Introduction
- Removal of visible contaminants such as oil, grease, dust, dirt, drawing compounds, and cutting compounds is necessary in any coating project.
- Solvent cleaning is a form of surface preparation that uses chemical solutions to remove visible contaminants from a surface.
▶️ Click here to understand in details about Solvent Cleaning process.
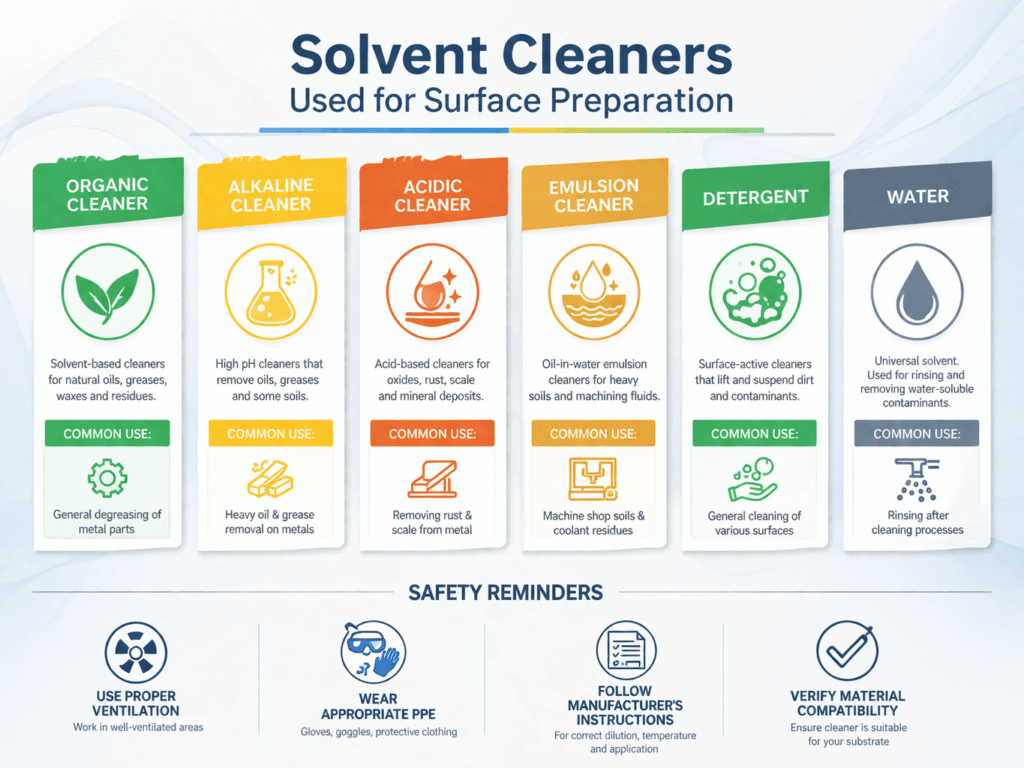
Solvent Cleaners
- There is a wide range of different solvent cleaners available. In the coatings industry today, pure organic solvents are rarely used to remove contaminants from the surface.
- They are being replaced by more environmentally friendly cleaners, such as detergents and emulsifying cleaners.
- Acceptable solvent types that conform to SSPC-SP 1 or ISO 8504 include organic solvents, detergents, emulsifiers, water, steam, alkaline cleaners, and acidic cleaners.
- The compatibility of the solvent cleaner and existing coating needs to be assured, as chemicals contained within the selected solvent may not be compatible with the coating specified for the project
1. Organic Solvents:
- Include kerosene, naphtha, turpentine, mineral spirits, toluene, and xylene.
- Cleans metal by dissolving and diluting the oil and grease contamination on the surface – Will leave behind a contaminate layer that interferes with adhesion.
- Cannot remove inorganic materials, such as chlorides, sulfates, weld flux, and mill scale.
- Can dissolve surface contaminants causing them to contaminate the surface and can dissolve some paint films. VOC and HAP’s regulations often prohibit the use of organic solvents as cleaners in many areas
2. Alkaline Cleaners:
- Include trisodium phosphate (TSP), sodium hydroxide, wetting agents, and soaps.
- Clean by wetting, emulsifying, dispersing, and dissolving containments.
- Multiple rinse cycles may be necessary after cleaning
Note: Emulsify – means to suspend a liquid in another liquid or to create an emulsion or smooth liquid from two liquids
3. Acidic Cleaners:
- Are typically composed of fairly strong acids, such as phosphoric acid (H3PO4), with small quantities of surfactants, water-miscible solvents, and organic wetting emulsifying agents.
- Limited effectiveness on removing grease/oil from surfaces. Very effective at removing mill scale and rust from steel.
- Commonly used, effective method of inexpensively roughening steel and concrete surfaces as a substitute for abrasive blast cleaning.
4. Detergents:
- Are typically composed of water-soluble buffering salts, dispersants, inhibiting agents, and soaps.
- Work by wetting, emulsifying, dispersing the contaminants, which are then washed away with hot water or steam.
- Are being increasingly used (especially the biodegradable types) due to the safety considerations associated with using alkaline or acidic cleaners
5. Emulsion Cleaners:
- Are typically composed of water-soluble soaps or emulsifying agents, buffering salts, dispersants, and inhibitors, along with mild organic dispersions to enhance cleaning/removal properties.
- Are formulated to spray onto the surface where they, like detergents, wet, emulsify, disperse, and float the contaminants to the surface of the substrate.
- Assist contaminants to rise to the surface allowing easy removal by vacuuming, pressure washing, or steam cleaning
6. Fresh Water (Used in Conjunction with Steam Cleaning or Pressure Washing):
- Water by itself is a powerful solvent cleaner that can effectively clean surfaces.
- Require testing of the water for pH, contaminants, and conductivity before being used for surface preparation.
– Steam cleaning (with or without a cleaning compound or detergent),
pressure washing (with or without a cleaning compound or detergent)
Solvent Cleaning: Safety Considerations
- Care must be taken during the handling, use, and disposal of solvents, especially when working with toxic solvents.
- Inspectors need to read the Product and Safety Data Sheets for any solvent cleaner used during the project.
- Workers may be required to complete special training prior to handling these chemicals.

1. Personal Protective Equipment (PPE) :
- PPE is essential for the safety of the individuals’ pre-cleaning surfaces, in addition to any other workers in the area.
- This includes the coating inspector. Workers should wear rubber gloves, safety goggles or eyeglasses, and respirators when applying or working near these materials.
- Solvents can release hazardous fumes that can be inhaled. As such, the concentration of the fumes should be below threshold limit values (TLV) defined by federal, state, or local regulations.
- Appropriate respiratory protection equipment should be used whenever the safe concentration is exceeded and when working in confined spaces.
2. Flash Points and Ignition Hazards :
- The flash point of some solvents is relatively low and can pose a significant jobsite hazard.
- The flash point is the temperature at which there is sufficient (solvent) vapor to ignite if a spark is generated causing a fire to form or an explosion to occur.
- High concentrations are most likely to occur in confined spaces, such as tanks, pipes, or vessels.
- Solvent concentration in the air should be monitored and never exceed the lower limit of flammability found on the safety data sheet.
- The lower limit of flammability is also known as the lower explosive limit (LEL). Other safety precautions include proper ventilation, no smoking or open flames, proper equipment grounding, and the use of appropriate personal protective equipment.
Disposal :
- Solvents must be contained and disposed of properly to not cause damage to the environment.
- The exact requirements will depend on the type of solvent being used as well as vary greatly from one county, state, and country to another.
- For example, when using alkaline or acidic cleaners, workers cannot wash contaminants onto the ground, into the normal drainage system, into adjacent watercourses, or allow the contaminants to run off into the general water system. Special collection and containment systems need to be in place for disposal purposes.
- In addition to the solvents themselves, any equipment used such as rags should also be disposed of correctly. Oil-soaked rags are a spontaneous combustion hazard because the oil can oxidize, release heat and ignite.

